Switching your child from liquid medicine to chewable tablets can feel like a huge leap. One minute you are carefully measuring milliliters with an orange spoon, and the next, you are handing them a small, often brightly colored disc. For many parents, this transition is a welcome relief. It means no more sticky spills on the kitchen counter, fewer arguments over taste, and easier packing for school trips. However, moving from a liquid suspension to a solid form isn't just about swapping containers. It involves understanding how the body absorbs different forms of medicine and ensuring the dose remains accurate.
If done correctly, transitioning to chewable tablets can improve medication adherence significantly. Studies show that non-compliance rates for pediatric liquid medications hover between 40% and 60%, largely due to taste aversion and administration difficulties. Chewables solve many of these problems, but they come with their own set of rules. You cannot simply guess the equivalent dose; you must calculate it based on weight and specific product strengths. This guide breaks down exactly how to make that switch safely, accurately, and without stress.
Why Make the Switch?
Before diving into the math, it helps to understand why manufacturers and doctors recommend chewable formulations. The primary driver is convenience combined with stability. Liquid medications, especially antibiotics and certain pain relievers, often require refrigeration. According to data from the American Association of Pharmaceutical Scientists, nearly 68% of pediatric liquids need to be kept cold to maintain potency. Chewable tablets, on the other hand, are stable at room temperature (typically 25°C) and have a shelf life of 24 to 36 months, compared to just 12 to 18 months for most suspensions.
Dosing accuracy is another major factor. When you pour liquid medicine into a cup, human error creeps in. Tilting the bottle too far, reading the meniscus incorrectly, or using a standard teaspoon instead of a calibrated syringe can lead to dosing errors of 15% to 20%. In contrast, a chewable tablet provides a fixed, pre-measured dose. If the label says 160mg, that is exactly what your child receives. This precision reduces the risk of under-dosing, which can lead to treatment failure, or over-dosing, which poses safety risks.
Portability also plays a role. If your child attends daycare or goes on family vacations, carrying a bulky plastic bottle that might leak is cumbersome. A small blister pack of tablets fits easily in a backpack. For older children, particularly those aged 6 to 12, taking a pill can also be a step toward independence, helping them manage their own health care as they grow.
Understanding Dosage Equivalence
The biggest mistake parents make when switching forms is assuming that one milliliter of liquid equals one tablet. This is rarely true. To convert safely, you must look at the active ingredient concentration in both products. Start by checking the label of the current liquid medication. Note the strength, usually expressed as milligrams per milliliter (e.g., 160mg/5mL). Then, check the strength of the chewable tablet (e.g., 160mg per tablet).
In this example, 5mL of liquid contains 160mg, and one tablet contains 160mg. Therefore, 5mL of liquid is equivalent to one tablet. But what if the numbers don’t match perfectly? Suppose the liquid is 80mg/5mL and the tablet is 160mg. You would need two doses of the liquid (10mL total) to equal one tablet. Always perform this calculation before buying the new medication. If the math results in a fraction of a tablet (like half a tablet), verify with your pharmacist whether splitting the tablet is safe. Not all chewables can be split evenly without affecting the dose distribution.
Weight-based dosing is critical here. Pediatric doses are calculated based on the child’s body weight, not just age. Use a reliable calculator or consult your doctor to determine the exact milligram amount needed per dose. Once you have that target number, find the chewable tablet strength that gets you closest to that number without exceeding it. Never round up arbitrarily. If your child needs 120mg and the only available chewable is 160mg, do not give the full tablet unless instructed otherwise. Instead, ask your pharmacist about alternative strengths or formulations.
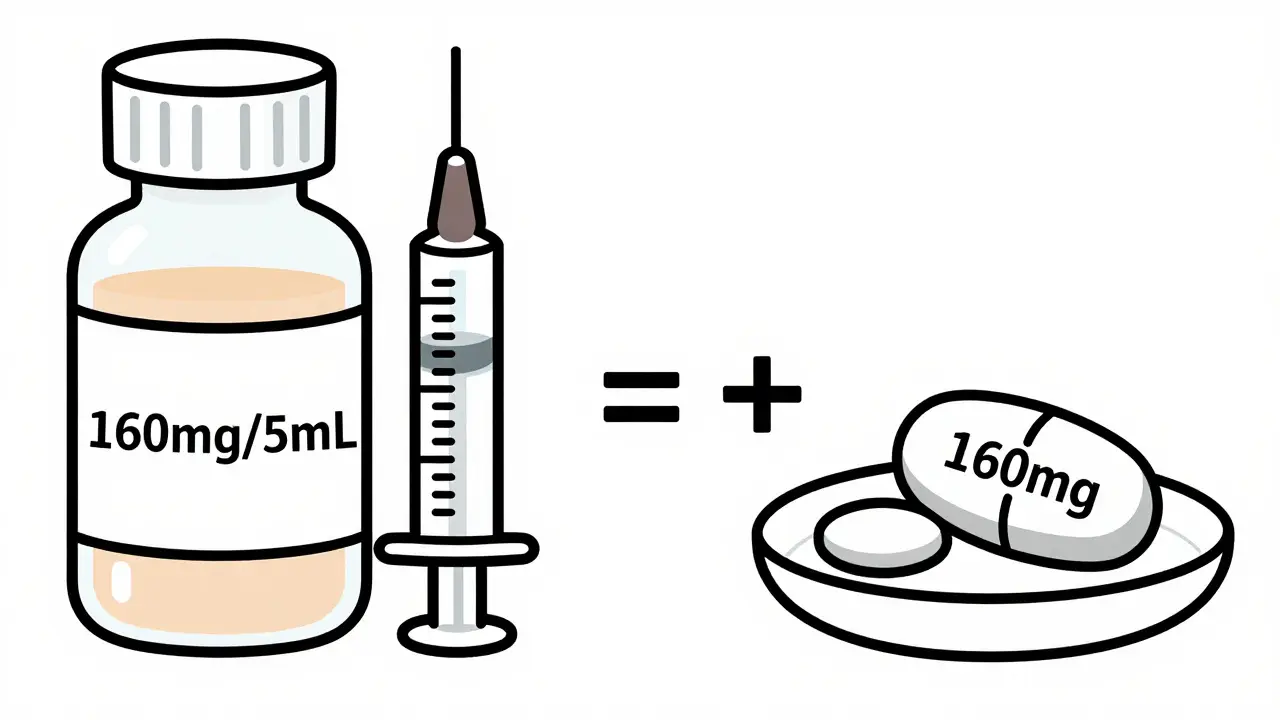
Step-by-Step Transition Process
Making the switch smoothly requires a structured approach. Do not abruptly stop the liquid and start the tablet on the same day without preparation. Follow these steps to ensure success:
- Verify Bioequivalence: Confirm with your pharmacist that the chewable version has similar bioavailability to the liquid. Most immediate-release chewables meet FDA standards for disintegration and dissolution, meaning they release the drug at a comparable rate. However, some extended-release formulations behave differently and should not be substituted casually.
- Assess Chewing Ability: Ensure your child can chew thoroughly. Children under two years old generally lack the motor skills to break down tablets effectively. For ages 2 to 12, observe them chewing hard foods like apples or raw carrots. If they tend to gulp food whole, they may swallow the tablet intact, which defeats the purpose of a chewable formulation.
- Educate on Technique: Teach your child to chew the tablet for at least 15 to 20 seconds. Research from University College London indicates that inadequate chewing leads to larger particle sizes, which can slow down drug absorption. Explain that the tablet needs to turn into a soft mush before swallowing.
- Start with a Trial Dose: Administer the first few doses under supervision. Watch for signs of difficulty, such as gagging or coughing. If your child struggles, try crushing the tablet and mixing it with a small amount of soft food like yogurt or applesauce, provided the manufacturer allows this.
- Monitor for Effectiveness: Keep a log for the first week. Note any changes in symptom control. If a fever persists longer than expected or pain returns quickly, the absorption rate might differ slightly, or the dose may need adjustment. Contact your healthcare provider if you notice significant changes.
Safety Precautions and Common Pitfalls
While chewable tablets are safer in many ways, they introduce new risks. The most common issue is accidental swallowing. Unlike regular tablets designed to dissolve slowly in the stomach, chewables rely on mechanical breakdown in the mouth to increase surface area for faster absorption. If swallowed whole, some chewables-especially those lacking strong disintegrants-can take over two hours to dissolve. This delay can reduce therapeutic effectiveness, potentially leading to breakthrough symptoms.
To mitigate this, choose products with clear instructions. Some brands, like Pfizer’s Advil Children’s Chewables, are formulated to be palatable and easy to break down. Others, particularly antacids like Tums, must be chewed thoroughly to work properly. Read the package insert carefully. Look for warnings about "do not swallow whole."
Allergies and sensitivities are another concern. Chewable tablets often contain additional excipients such as sugars, artificial flavors, or dyes to mask the bitter taste of the active ingredient. Children with diabetes or sugar-phobia should avoid high-sugar formulations. Check for corn starch or gluten if your child has specific allergies. Microcrystalline cellulose and hydroxypropyl methylcellulose are common binders that are generally safe, but always review the inactive ingredients list.
Storage conditions matter too. While chewables are more stable than liquids, they are still sensitive to moisture. Store them in a cool, dry place away from bathrooms where humidity fluctuates. Moisture can cause tablets to crumble or degrade prematurely. Keep them in their original blister packs until use to protect against environmental factors.
| Feature | Liquid Suspension | Chewable Tablet |
|---|---|---|
| Dosing Accuracy | Variable (15-20% error potential) | High (<2% variance) |
| Shelf Life | 12-18 months | 24-36 months |
| Storage Requirements | Often requires refrigeration | Room temperature stable |
| Absorption Speed | Faster (pre-dissolved) | Slightly slower (requires mastication) |
| Taste Masking | Good, but can leave aftertaste | Excellent, sweet/flavored options |
| Portability | Bulky, leak-prone | Compact, travel-friendly |

When Not to Switch
Despite the benefits, chewable tablets are not suitable for every situation. Avoid switching if your child has severe dysphagia (difficulty swallowing) or neurological conditions that impair chewing coordination. In these cases, liquid formulations remain the safest option because they require minimal oral manipulation.
Also, consider the urgency of treatment. Liquids absorb faster because they are already dissolved. For acute emergencies, such as severe allergic reactions or high fevers requiring rapid reduction, liquids may act 15 to 30 minutes faster than chewables. If speed is critical, stick with the liquid form.
Finally, cost and availability can be barriers. Not all medications have chewable equivalents. Some specialty drugs or newer antibiotics are only available in liquid or capsule form. Always check with your pharmacy before attempting to substitute. Never crush or split capsules or extended-release tablets, as this can release the entire dose at once, causing toxicity.
Tips for Success
Transitioning successfully often comes down to behavioral strategies. Make the process positive. Let your child pick the flavor if multiple options are available. Use praise and rewards for successful chewing. Avoid forcing the tablet if resistance is high; pause and try again later or revert to liquid temporarily.
Pair the medication with a consistent routine. Giving the tablet after breakfast or before bed creates a habit loop. Consistency helps children adapt to the new texture and taste. Over time, the novelty wears off, and it becomes a normal part of their day.
If your child complains about the size of the tablet, know that you have options. Many manufacturers offer lower-strength versions that are smaller in diameter. Alternatively, ask your pharmacist about compounding services, which can create custom-sized tablets or flavored powders. Innovation in pharmaceuticals is also bringing forth "intelligent" chewables that change color when adequately chewed, providing visual feedback for proper administration.
Can I crush a chewable tablet if my child refuses to chew it?
Generally, yes. Since chewable tablets are designed to be broken down, crushing them is usually safe. Mix the powder with a small amount of soft food like applesauce, yogurt, or pudding. However, always check the label first. Some medications may taste extremely bitter when crushed, which could deter your child. Also, ensure the medication is not an extended-release formulation, as crushing those can be dangerous.
At what age can children start taking chewable tablets?
Most experts recommend starting around age 2 to 3, provided the child has developed adequate chewing skills. Younger toddlers may lack the motor control to break down the tablet fully, increasing the risk of choking or ineffective absorption. For children under 2, liquid suspensions are typically safer and more effective. Always consult your pediatrician before introducing solid forms to very young children.
What if my child swallows the tablet whole?
If your child swallows a chewable tablet whole, it may not dissolve quickly enough to provide the intended therapeutic effect. This can lead to reduced efficacy or delayed symptom relief. Monitor your child closely for signs that the medication is working. If symptoms persist, contact your healthcare provider. They may advise giving an additional dose or switching back to liquid form temporarily. Teaching proper chewing technique is essential to prevent this.
Do chewable tablets expire faster than liquids?
No, chewable tablets typically have a longer shelf life. While liquid medications often expire within 12 to 18 months, especially after reconstitution, chewable tablets can last 24 to 36 months when stored properly at room temperature. Their solid form makes them less susceptible to microbial growth and chemical degradation. Always check the expiration date on the packaging before use.
Is the dose of a chewable tablet always the same as the liquid?
Not necessarily. You must calculate the equivalence based on the active ingredient concentration. For example, 5mL of liquid containing 160mg is equivalent to one 160mg tablet. However, different brands may have varying strengths. Always compare the milligram amounts rather than assuming volume equals count. Consult your pharmacist to ensure the converted dose matches your child’s weight-based requirements.

All Comments
Srinivas Komakula May 5, 2026
The pharmaceutical industry has long manipulated the public perception of dosage accuracy to push their proprietary formulations. You cannot trust these so-called "stable" chewables without questioning the excipients used. The mention of microcrystalline cellulose is a red flag for those who understand the hidden agendas of Big Pharma. They claim precision, yet they ignore the systemic impact of artificial dyes on developing neurological systems. The FDA standards cited here are merely guidelines designed to protect corporate profits rather than patient health. One must scrutinize every ingredient list with extreme skepticism. The narrative of convenience is a distraction from the potential toxicity of binders and preservatives. Do not fall for the illusion of safety provided by brightly colored discs. These products are engineered for compliance, not necessarily for optimal physiological outcomes. Always verify the raw chemical composition before administering anything to your child.
Preety Singh May 6, 2026
This article lacks the sophistication expected of modern parenting literature. The advice given is rudimentary at best. One would assume that educated parents already understand the basics of pharmacokinetics. The suggestion to simply "chew thoroughly" ignores the complex motor skill development required in early childhood. Furthermore, the reliance on manufacturer labels is naive. True expertise lies in understanding the molecular structure of the active ingredients. Most readers will likely skim this content without grasping the deeper implications. It is disappointing to see such basic information presented as groundbreaking. A truly informed parent would consult a specialist rather than reading generic online guides. The distinction between immediate-release and extended-release formulations requires a level of knowledge beyond this text. Only the most diligent caregivers will find value in these superficial observations.
Seema Karanje May 6, 2026
Stop wasting time debating the merits of liquids versus tablets! Just get it done! Your child needs medication now, not a lecture on shelf life. If you are too lazy to calculate the dose properly, then you are failing as a parent. The math is simple; do not make excuses. Chew the tablet or crush it; whatever works fastest. Do not let fear paralyze you into inaction. Procrastination is the enemy of health. Get up, go to the pharmacy, and buy the right product. Then administer it correctly without hesitation. Confidence is key here. If you hesitate, your child will sense your uncertainty. Take charge of the situation immediately. There is no time for second-guessing when it comes to health. Act decisively and move forward.
J. Walter Jenkem May 8, 2026
I appreciate the collaborative tone of this guide. It feels like we are all working together to improve pediatric care practices. The step-by-step approach makes the transition feel manageable for everyone involved. I think focusing on education rather than fear is the right path forward. Let us support each other in ensuring our children receive accurate doses. The community aspect of sharing experiences can only help reduce anxiety. We should encourage open dialogue with healthcare providers about these options. Together, we can create a safer environment for medication administration. Thank you for fostering this sense of unity among parents. It is important to remember that we are not alone in this journey.
Mark Koepsell May 10, 2026
The technical details regarding disintegration rates are crucial for understanding efficacy. Many users fail to realize that inadequate chewing significantly alters absorption profiles. The reference to University College London research adds necessary credibility to the claims. It is essential to distinguish between different formulation types before switching. Bioequivalence is not guaranteed across all brands. Pharmacists play a vital role in verifying these parameters. Users should document any changes in symptom control meticulously. This data can be invaluable for future medical consultations. Precision in administration leads to better therapeutic outcomes. Always prioritize verified information over anecdotal evidence.
Elizabeth Holden May 11, 2026
omg this is so basic lol. like really? you need a whole article for this?? just read the label dummy. its not rocket science. why do people make everything so complicated. my kid chews his vitamins fine so he can handle meds. dont overthink it. if you cant figure out half a tablet you are doing it wrong. seriously though this is obvious stuff. waste of my time reading all this fluff. just give the pill and move on. nobody cares about the fancy words here. keep it simple folks.
Jenny X May 12, 2026
The establishment wants you to believe that chewables are safer because they control the supply chain more effectively. Look at the shelf life extension; it is clearly a strategy to increase inventory turnover while reducing cold storage costs for distributors. The mention of "human error" in liquid dosing is a convenient scapegoat for poor manufacturing quality control. They want you dependent on their fixed-dose units. Never forget that the FDA is funded by the very companies producing these tablets. The narrative of independence for older children is a psychological manipulation tactic. Children are being conditioned to accept solid pills as a rite of passage. Question the motives behind every recommendation. The truth is hidden in the inactive ingredients list.
bharat films May 13, 2026
Great points on the moisture sensitivity! 😊 Tablets can crumble if stored incorrectly. 📦 Always keep them dry! 💧 The comparison table was very helpful. 📊 I learned something new today. 🎉 Thanks for sharing this info. 👍 It helps parents make better choices. 🌟 Stay safe everyone! 🛡️
Mikaela -anonymous 😏 May 15, 2026
Oh, look at us, playing house with little plastic discs. How quaint. The idea that we can simply "swap containers" is laughably naive. Life is not a video game where you press a button and the problem disappears. You are gambling with your child's health based on a blog post. The arrogance of assuming one size fits all is breathtaking. And don't get me started on the "independence" angle. That is just code for less parental supervision. Enjoy your breakthrough symptoms when the tablet fails to dissolve. At least you tried. Sarcasm aside, please check with a doctor. But honestly, who reads the fine print anyway?
kelvin villa saab May 15, 2026
this is totaly wrong u guys are idiots. i told my kid to swallow it whole and he was fine. why do u need to chew it?? it goes down same way. ur article is bs. dont listen to them. just throw it in mouth and drink water. easy peasy. stop complicating things. u are all crazy for worrying about digestion rates. it works either way. trust me i know what im talking about even tho im not a doctor. lol bye.