Tag: bioequivalence
How to Switch Back from Generic to Brand Name Medication Safely
- Cheryl Moran
- May 2, 2026
- 0 Comments
Learn how to safely switch from generic to brand-name medication. We cover bioequivalence, prior authorization steps, insurance denials, and when a switch is medically necessary.
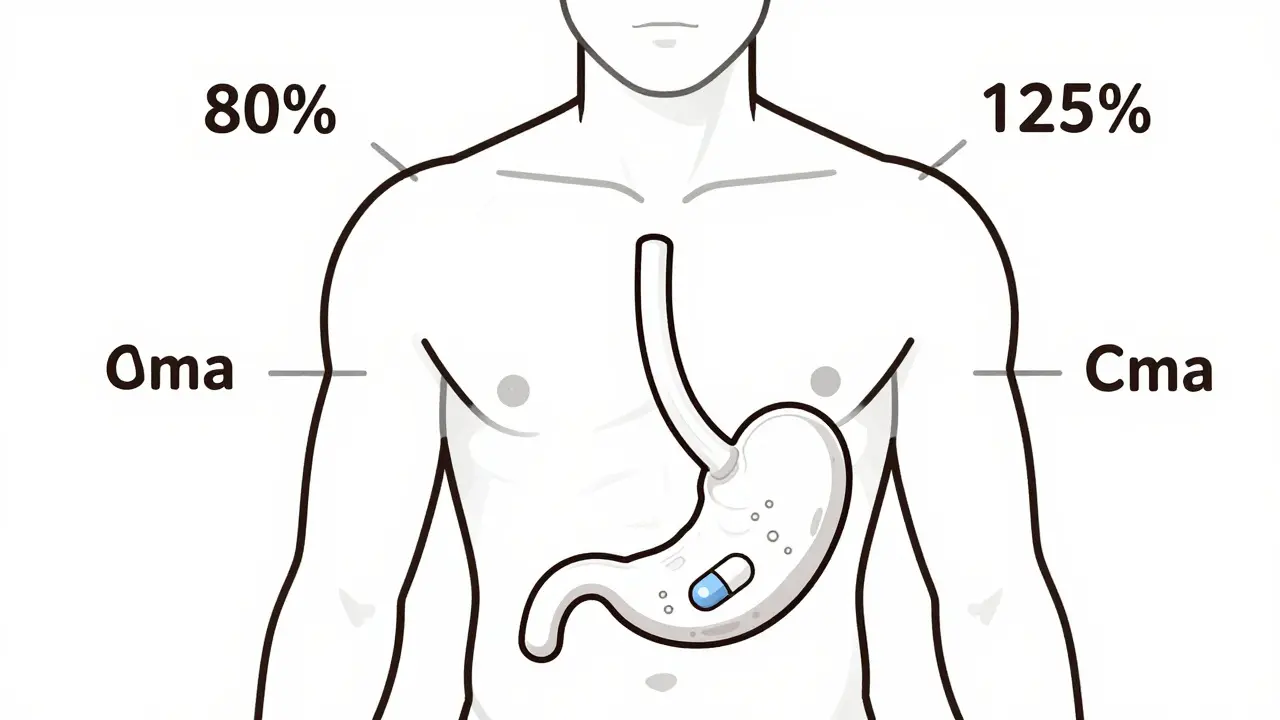
read moreThe 80-125% Rule: Understanding Bioequivalence Confidence Intervals in Generic Drugs
- Cheryl Moran
- January 27, 2026
- 12 Comments
The 80-125% rule ensures generic drugs work like brand-name versions by measuring how much drug enters your bloodstream. It's not about pill strength-it's about body absorption. Here's how it works.
read more